Safic-Alcan is a global specialty chemicals distributor providing direct access to a wide and innovative product portfolio. Beyond distribution, our regulatory experts support partners worldwide to ensure compliance with REACH regulations while anticipating future regulatory and industry trends.
One key element of REACH is the SVHC product list, which plays a major role in substance management and compliance.
What does SVHC mean?
SVHC stands for Substances of Very High Concern. These substances may have serious effects on human health or the environment. Under REACH, a substance can be identified as SVHC if it falls into one of the following categories:
- Carcinogenic (category 1A or 1B)
- Mutagenic (category 1A or 1B)
- Toxic to reproduction (category 1A or 1B)
- Persistent, bioaccumulative and toxic (PBT)
- Very persistent and very bioaccumulative (vPvB)
- Substances of equivalent concern (e.g. endocrine disruptors or respiratory sensitisers)
Who decides if a chemical is an SVHC?
EU Member States, or the European Chemicals Agency (ECHA) at the request of the European Commission, can propose a substance as an SVHC. This is done by submitting a dossier in line with Annex XV of REACH.
Before submission, the intention is published in the Registry of Intentions, allowing stakeholders to prepare. Once the proposal is published, a 45-day public consultation is opened. During this period, companies and interested parties can submit comments, data, or information on uses and alternatives.
If new information is provided or the proposal is challenged, the case is reviewed by the Member State Committee (MSC).
- If agreement is unanimous, the substance is added to the Candidate List
- If not, the final decision is taken by the European Commission
Safic-Alcan strongly encourages companies to participate in public consultations, ensuring decisions are based on the most complete and accurate data available.
When is the SVHC list updated?
The SVHC Candidate List is updated twice a year, typically in January and July.
As of the last update (10 June 2022), the list contains 224 substances.
The official list is available on the ECHA website.
Am I concerned by the SVHC list?
If your company manufactures, imports, or sells substances, mixtures, or articles in the EU, you may have legal obligations as soon as a substance is added to the Candidate List.
These obligations apply to:
- Substances on their own
- Substances in mixtures
- Substances present in articles
Definitions of substances, mixtures, and articles can be found in Article 3 of REACH.
Safic-Alcan advises companies to clearly identify whether their product is a substance, mixture, or article under REACH.
What are the obligations related to SVHCs?
Depending on your product and its composition, obligations may include:
- Informing customers and consumers
- Notifying ECHA
- Submitting information to the SCIP database
- Providing Safety Data Sheets (SDS)
- Minimising environmental releases
Detailed requirements are available on ECHA’s dedicated pages.
Important reminder: As long as an SVHC is not included in the Authorisation List (Annex XIV of REACH), its use is not prohibited.
What is the link between SVHC and Authorisation?
The authorisation process aims to progressively replace SVHCs with safer alternatives when technically and economically feasible.
ECHA regularly reviews substances on the Candidate List to decide which should be prioritised for inclusion in the Authorisation List. This decision is based on:
- Hazard properties
- Volume placed on the market
- Wide dispersive uses
A three-month public consultation is launched as part of this process.
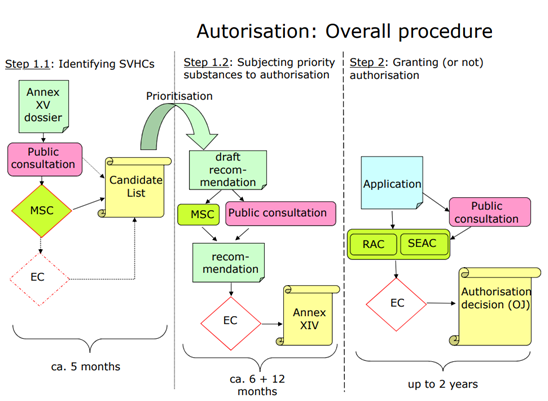
What happens if a substance is added to the Authorisation List?
To continue using a substance after its sunset date, companies must:
- Submit an application for authorisation
- Do so before the latest application date
- Pay the associated fees
Standard ECHA fees include:
- Base fee: EUR 54,100
- Additional fee per substance: EUR 10,820
- Additional fee per use: EUR 48,690
Safic-Alcan recommends starting substitution efforts as soon as a substance appears on the SVHC list, as the ultimate goal is to phase out these substances.
Sources :
Glossary :
- CSR: Chemical safety report
- ECHA: European Chemicals Agency
- EEA: The European Economic Area
- EU: European Union
- MSC: Member State Committee
- PBT: persistent, bioaccumulative and toxic
- REACH: Registration, Evaluation and Authorisation of Chemicals
- SCIP: Substances of Concern In articles as such or in complex objects (Products)
- SVHC: Substance of Very High Concern
- UVCB (unknown or variable composition, complex reaction products or of biological materials)
- vPvB: very persistent and very bioaccumulative
- WFD: Waste Framework Directive
