TL;DR
Three major changes affect Annex V (permitted preservatives) in 2026:
- 1 May 2026 — Commission Regulation (EU) 2026/78 applies: new concentration limits for o-Phenylphenol and Sodium o-Phenylphenate, new bans on CMR substances. No transitional period.
- 15 July 2026 — New mandatory labelling for formaldehyde-releasing preservatives: warning required when released formaldehyde exceeds 0.001% (10 ppm).
- 31 July 2026 — Full compliance deadline for fragrance allergen labelling under Regulation (EU) 2023/1545 (80 allergens, Annex III).
Ready to Work Together?
Get in touch with our team and discover tailored solutions for your industry
What Is Annex V of the EU Cosmetics Regulation?
Annex V of Regulation (EC) No 1223/2009 is the list of permitted preservatives in cosmetic products sold within the European Union and EEA.
For each listed substance, Annex V specifies:
- Name and INCI designation — chemical name and CAS/EC number
- Maximum concentration — per product type (rinse-off vs. leave-on)
- Scope restrictions — product categories, application routes, age groups
- Mandatory label warnings — where required for consumer safety
Preservatives not listed in Annex V cannot be used as such in EU cosmetic products. Annex V is updated regularly as the Scientific Committee on Consumer Safety (SCCS) issues new opinions and as CMR classifications under the CLP Regulation are updated.
The Key Driver in 2026: CMR Reclassifications Under the CLP Regulation
Most 2026 changes to Annex V are driven by a single upstream event: Commission Delegated Regulation (EU) 2024/2564 — the 21st Adaptation to Technical Progress (ATP) of the CLP Regulation — which updated the harmonised classification of several substances as CMR (carcinogenic, mutagenic, or reprotoxic).
Under Article 15 of Regulation (EC) No 1223/2009, CMR substances of category 1A, 1B, or 2 are prohibited in cosmetics by default, unless the SCCS has assessed them as safe under specific conditions of use.
This mechanism directly triggered Commission Regulation (EU) 2026/78, published on 13 January 2026, which amends Annexes II, III, IV, and V of the Cosmetics Regulation.
Application date: 1 May 2026. No transitional period.
Understanding the CLP-to-Cosmetics pipeline is essential for anticipating future restrictions. Safic-Alcan's regulatory team monitors these classifications upstream — for more on how REACH and SVHC classifications interact with cosmetic compliance obligations, see our dedicated article.
Annex V Changes Effective 1 May 2026
This is the most significant Annex V change in 2026.
Background: o-Phenylphenol (CAS 90-43-7, INCI: o-Phenylphenol) has been listed in Annex V, entry 7, as a permitted preservative for rinse-off and leave-on products. It was reclassified as a CMR category 2 carcinogen by Delegated Regulation (EU) 2024/2564.SCCS assessment: The SCCS issued its opinion on 25 October 2024 concluding that both o-Phenylphenol and its sodium salt (Sodium o-Phenylphenate, CAS 132-27-4) can be considered safe under specific conditions.
What changes in entry 7 from 1 May 2026:
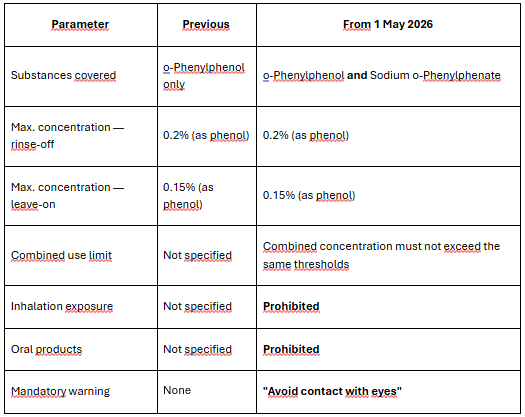
Key formulation implications:
- Products using o-Phenylphenol alone: update CPSR and labelling to reflect new warning and use restrictions.
- Products using both substances: verify combined concentration compliance.
- Spray or aerosol formats: assess inhalation exposure risk — reformulation may be required.
- Oral care products (mouthwash, toothpaste): these preservatives are now prohibited.
New CMR Substance Bans Affecting Annex V
Regulation (EU) 2026/78 also adds several CMR substances to Annex II (prohibited substances). Among those relevant to preservation systems:
- Trimethyl borate (CAS 121-43-7) — prohibited from 1 May 2026, no adaptation period.
- N,N'-methylenediacrylamide (CAS 110-26-9) — prohibited from 1 May 2026.
These bans apply regardless of concentration. Any product containing these substances must be withdrawn from the EU market by 1 May 2026. There is no sell-through period.
Annex V Preamble Change: Formaldehyde-Releasing Preservatives
This change does not originate from Regulation (EU) 2026/78 — it stems from Commission Regulation (EU) 2022/1181, which amended the preamble of Annex V.
What changes: The threshold triggering the mandatory formaldehyde warning is reduced from 0.05% (500 ppm) to 0.001% (10 ppm). New mandatory warning text (from 15 July 2026): "releases formaldehyde" — required on all finished products where the total concentration of formaldehyde released exceeds 0.001%, regardless of whether one or multiple formaldehyde-releasing substances are present.
Commonly affected preservatives in Annex V:
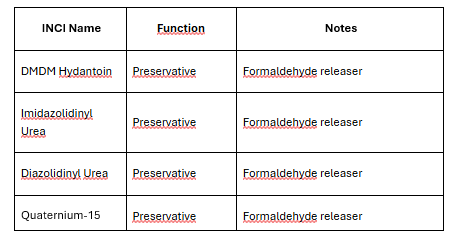
Why the threshold was lowered: The SCCS concluded that the previous 0.05% threshold was insufficient to protect consumers already sensitised to formaldehyde from developing contact dermatitis. This is a consumer safety-driven amendment, not a CMR reclassification.
Sell-through deadline: Products compliant with the old regulation (pre-July 2022) could be placed on the market until 31 July 2024, and made available until 31 July 2026. After this date, full compliance is mandatory.
For formulators using these preservatives, the practical question is binary: either add the new warning, or reformulate to bring released formaldehyde below 10 ppm. Safic-Alcan's cosmetics team can support the identification of alternative preservation systems — our article on cosmetic microbiology and preservation covers the technical principles and available solutions.
What Is Not Changing in Annex V in 2026
To avoid confusion, it is worth clarifying what is not affected by 2026 Annex V changes:
- Parabens — no new restrictions in 2026. Existing limits remain.
- Phenoxyethanol — no change in 2026. Still permitted at 1% maximum.
- Benzyl alcohol — no change in 2026.
- Chlorphenesin — no change in 2026.
These substances remain under ongoing SCCS review as part of the broader regulatory programme, but no application deadlines apply in 2026.
What Formulators and Regulatory Teams Should Do Now
1. Audit your preservation systems
Identify all products using o-Phenylphenol, Sodium o-Phenylphenate, formaldehyde-releasing preservatives, or any substance newly added to Annex II under EU 2026/78. Cross-reference against the updated annexes.
2. Update CPSRs
Any change in formulation, concentration, or use conditions requires an updated Cosmetic Product Safety Report. Ensure your safety assessor is aware of the new CMR classifications and SCCS opinions.
3. Revise labelling
- Add "Avoid contact with eyes" for products containing o-Phenylphenol or Sodium o-Phenylphenate.
- Add "releases formaldehyde" warning where applicable (threshold: 0.001%).
- Verify INCI nomenclature against the updated glossary (mandatory from 30 July 2026).
4. Assess spray and aerosol formats specifically
The prohibition of o-Phenylphenol in applications with inhalation exposure is new. Spray, mist, and aerosol formats using this preservative require immediate reformulation assessment.
5. Clear non-compliant stock
For substances banned under EU 2026/78 with no transitional period: products must not be placed on the EU market after 1 May 2026. There is no sell-through window.
FAQ
What is Annex V of the EU Cosmetics Regulation?
Annex V of Regulation (EC) No 1223/2009 is the list of preservatives permitted for use in cosmetic products sold in the EU and EEA. It specifies maximum concentrations, product type restrictions, and mandatory label warnings for each listed substance.
What is the most important Annex V change in 2026?
The amendment of entry 7 under Commission Regulation (EU) 2026/78, applicable from 1 May 2026. It adds Sodium o-Phenylphenate to the permitted preservatives list alongside o-Phenylphenol, introduces new concentration limits for combined use, prohibits use in products with inhalation exposure and in oral products, and requires the warning "Avoid contact with eyes".
Why are these changes happening?
Most 2026 changes are driven by updated CMR classifications under Commission Delegated Regulation (EU) 2024/2564 (21st ATP to the CLP Regulation). When a substance is reclassified as CMR, its use in cosmetics is automatically reviewed under Article 15 of the Cosmetics Regulation. The SCCS then assesses whether a safe use condition exists.
Does Regulation (EU) 2026/78 have a transitional period?
No. The regulation applies from 1 May 2026 with no adaptation period for the CMR substance bans. Products containing newly prohibited substances must be withdrawn from the EU market by that date.
Which formaldehyde-releasing preservatives are commonly used in cosmetics?
The most common are DMDM Hydantoin, Imidazolidinyl Urea, Diazolidinyl Urea, and Quaternium-15. All are listed in Annex V and are permitted, but products containing them must now carry the "releases formaldehyde" warning if released formaldehyde exceeds 0.001% (10 ppm) in the finished product — a deadline that applies from 15 July 2026.
Where can I find the official updated text of Annex V?
The consolidated version of Regulation (EC) No 1223/2009, including all annexes, is available on EUR-Lex. Commission Regulation (EU) 2026/78 is published in the Official Journal of the EU.
Is Safic-Alcan able to support reformulation in response to these changes?
Yes. Safic-Alcan's cosmetics team provides regulatory monitoring, ingredient sourcing, and formulation support across preservation systems. Contact our specialists for guidance on compliant alternatives and updated ingredient portfolios.
